
Due to the fact that most substances in drugs have hygroscopicity, a dry environment is crucial for drug production and storage. And during the production of drugs, the adsorption of moisture can affect the final weight and quality of the drugs.
A dry environment usually improves productivity, product quality, and drug shelf life. After years of practical experience, engineers from Tailixin have concluded that the pharmaceutical process requires a relative humidity control between 20% and 35%, and a temperature control of 23 ℃ ± 2. The soft capsule production line is a common production area in pharmaceutical enterprises with dehumidification requirements. The humidity requirement for the capsule drying workshop is generally 23 ± 2, with a relative humidity of 20%.
Medical products are sensitive products, such as powders, tablets, capsules, and diagnostic strips, which often lose their efficacy and shelf life when in contact with moisture in the air. These products will stick together, mold or break.
Simultaneously causing blockage of machines and pipelines, hindering production, transportation, and storage. This is an avoidable time and cost intensive situation. The key is to continuously supervise and monitor air humidity throughout all production processes, from raw materials to final products.
The drug production process in pharmaceutical factories is a guarantee of quality. Strengthening scientific supervision of drug production enterprises, standardizing various production processes, and controlling the quality of the entire production process plays a significant role in improving drug quality and reducing drug accidents. Tailixin rotary Dehumidifier is a special equipment for dehumidification, mold removal and environment improvement, which is designed to largely avoid the harm of moisture during drug production. Whether the production environment control in pharmaceutical factories is an effective tool and whether the production environment monitoring is qualified is a necessary condition for drug release.
It has been widely used in pharmaceutical factories, pharmaceutical workshops, pharmaceutical warehouses, and other places. The Dehumidifier uses the principle of freeze drying to suck in moist air, drop it below the dew point temperature through the evaporator, condense the moisture in the air into water drops, and then turn it into dry air after being heated up by the condensing heat of the refrigeration compressor, so as to achieve the purpose of drying air.


The desiccant Dehumidifier is usually placed in front of the rear surface cooling to remove water, so as to avoid condensation of the rear surface cooling, keep the air duct dry, and prevent bacterial growth. The effect of the Dehumidifier is controlled by the air volume passing the runner and the bypass ventilation valve. As more dry air is required in the room, the air volume passing the runner is more. When the temperature and humidity in the room meet the requirements, more air will bypass the dehumidification wheel through bypass method two. This design can maintain a relative humidity of ± 2% indoors. The typical case application of a pharmaceutical factory is shown in the following figure:
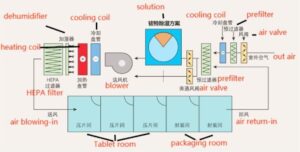
If your on-site working conditions are complex and require strict calculation to achieve the optimal temperature and humidity, you can also communicate with us for transportation.




